Menu
leadingthefashion
{{ variable.name }} : {{ variable.values[selectedVariantsIndex[variable_index]].name }}
Quantity:
| Parameter information | |
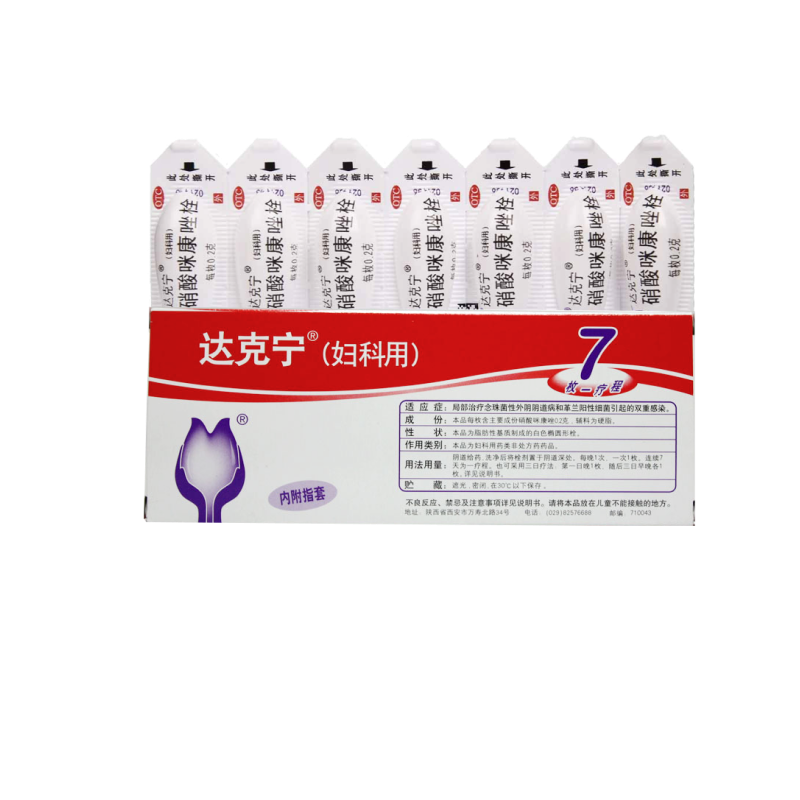
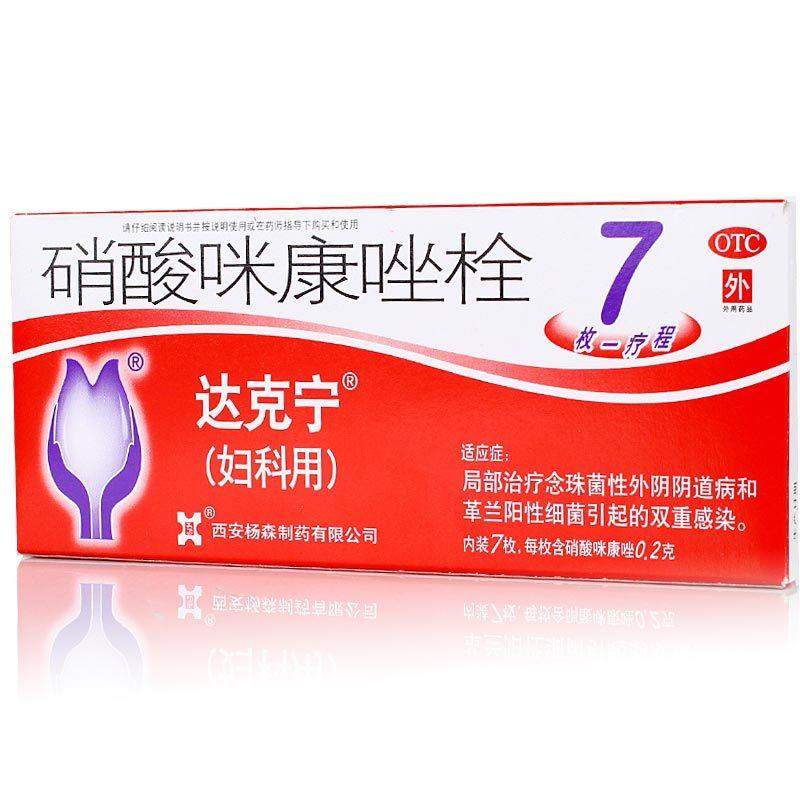
| Brand | Daktarin |
| Product Name | 0.2G*7 suppositories x 1 box, 0.2G*7 suppositories x 2 boxes, 0.2G*7 suppositories x 3 boxes, 0.2G*7 suppositories x 5 boxes |
| Manufacturing Company | Xi'an Janssen Pharmaceutical Co., Ltd. |
| Product Form | Suppository |
| Usage | Vaginal administration |
| Target Audience | Please refer to the instructions or consult healthcare professionals. |
| Specification | 0.2g*7 suppositories per box |
| Category | Chemical Drug |
| Approval Number | National Medicine Approval No. H10930214 |
| Drug Category | Over-the-counter (OTC) drug |
| Validity Period | 48 months |
| Dosage | Once every night, one suppository each time |
| Generic Name | Miconazole Nitrate Suppository |
| Drug Name | Miconazole Nitrate Suppository |
| Packaging Specification | 0.2G*7 suppositories |
| Indications | Candidal vulvovaginitis and mixed infections caused by Gram-positive bacteria |
| Symptoms | Candidal vulvovaginitis |
| Brand Name | Daktarin |