Menu
leadingthefashion
{{ variable.name }} : {{ variable.values[selectedVariantsIndex[variable_index]].name }}
Quantity:
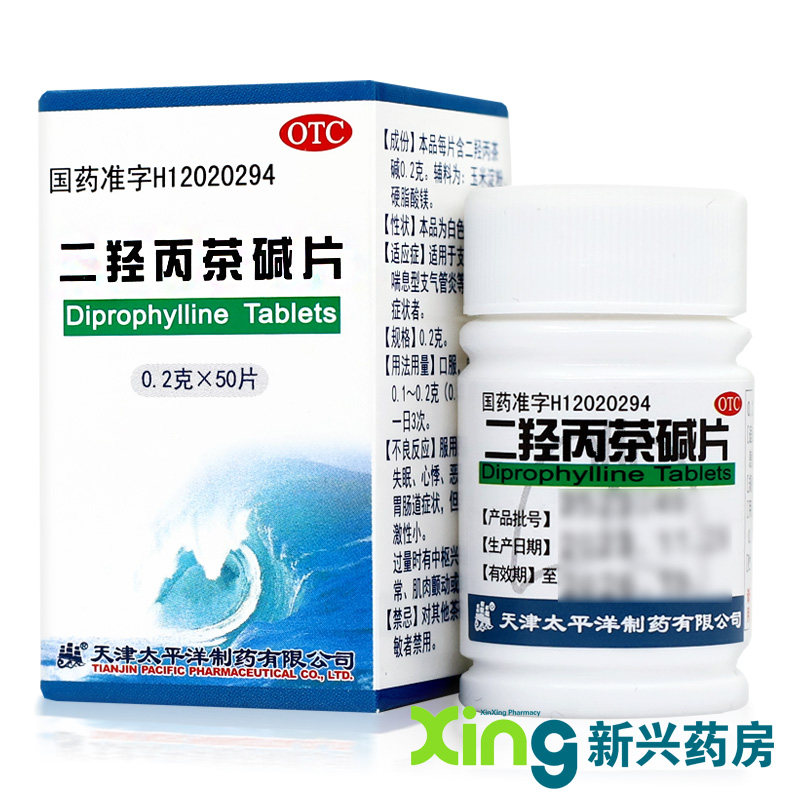




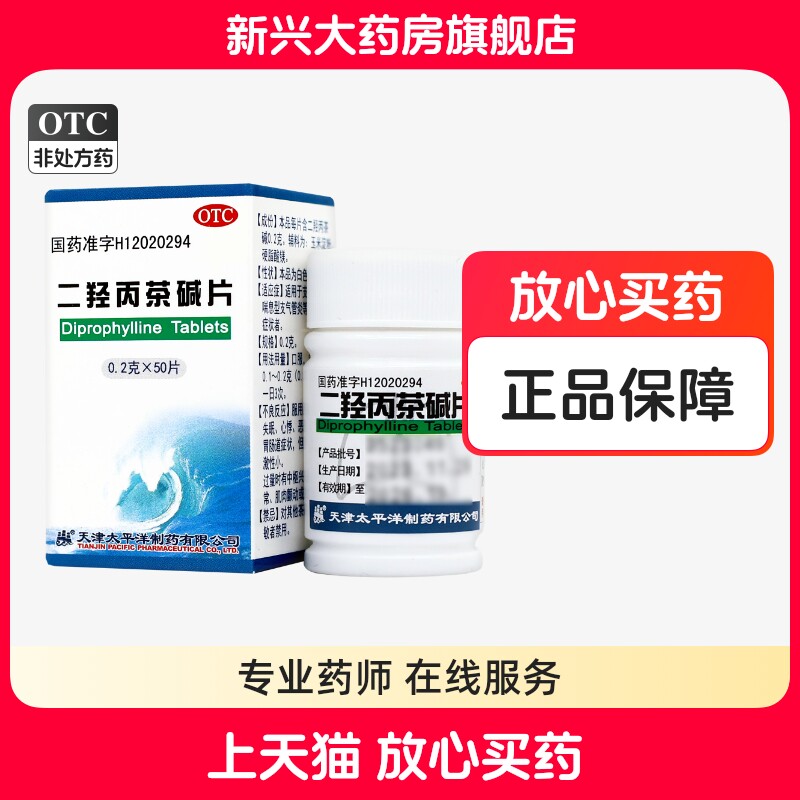
| Parameter information | |
| Brand | Pacific |
| Product Name | 50 tablets x 1 box |
| Manufacturing Company | Tianjin Pacific Pharmaceutical Co., Ltd. |
| Product Form | Tablet |
| Usage | Oral administration |
| Target Audience | Please refer to the instructions or consult healthcare professionals. |
| Specification | 0.2g x 50 tablets per bottle |
| Category | Chemical Drug |
| Approval Number | National Medicine Permit H12020294 |
| Drug Category | Over-the-counter (OTC) drug |
| Validity Period | 36 months |
| Dosage | Adults: 0.5 to 1 tablet per dose, three times daily |
| Generic Name | Doxofylline Tablets |
| Drug Name | Doxofylline Tablets |
| Packaging Specification | 50 tablets |
| Indications | Bronchial Asthma |
| Symptoms | Bronchial Asthma |